IND Strategy & Approval Readiness
IND Strategy & Approval Readiness
Integrated strategy design for IND approval readiness
Service Overview
Service Overview
IND approval is the first critical gateway in drug development and the pivotal stage that determines the success of subsequent clinical development.
LinkedBio designs integrated strategies encompassing non-clinical research, clinical trial planning, and overall development strategy to support high-probability IND submissions and approvals.
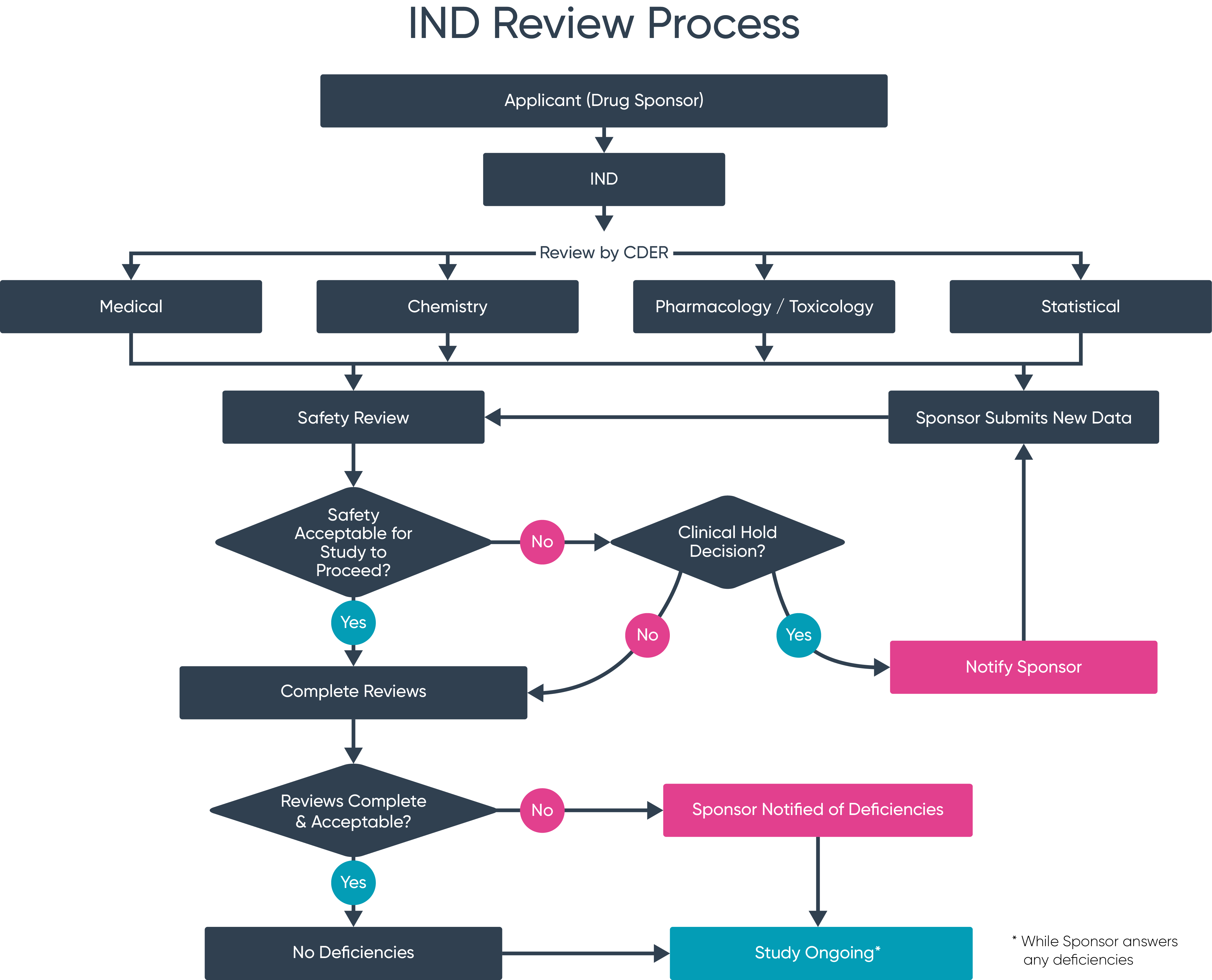
IND Submission Flow
IND Approval Process
Non-Clinical Review
Data Analysis
IND Strategy Design
Strategic Planning
Document Preparation
Dossier Assembly
IND Submission
Filing
Supplement Response
Deficiency Resolution
IND Approval
Clearance
Key Support Areas
Key Support Areas
Differentiators
Differentiators
01
Hands-on execution support and MFDS supplement/deficiency response — strategy development beyond consultant-only engagement
02
GAP analysis of non-clinical study results from a clinical-integration perspective
03
Full-lifecycle clinical trial development planning grounded in non-clinical study outcomes
04
IND strategy designed to connect with clinical trial operations
05
Strategic development planning to achieve IND approval